Imagine you are in a laboratory where scientists are developing systems to provide oxygen in extreme environments like submarines or space missions. In such cases, compounds like KO2 and KO3 play an important role. Understanding the difference between KO2 and KO3 is not only essential for chemistry students but also for professionals working in environmental science and industrial applications.
The difference between them lies mainly in their chemical composition, structure, and reactivity. While both are oxygen-rich compounds of potassium, they behave quite differently in practical situations.
Knowing the difference between both helps learners understand oxidation processes and oxygen generation systems more clearly. In this article, we will explore the difference between KO2 vs KO3 in a simple, structured, and engaging way.
Key Difference Between the Both
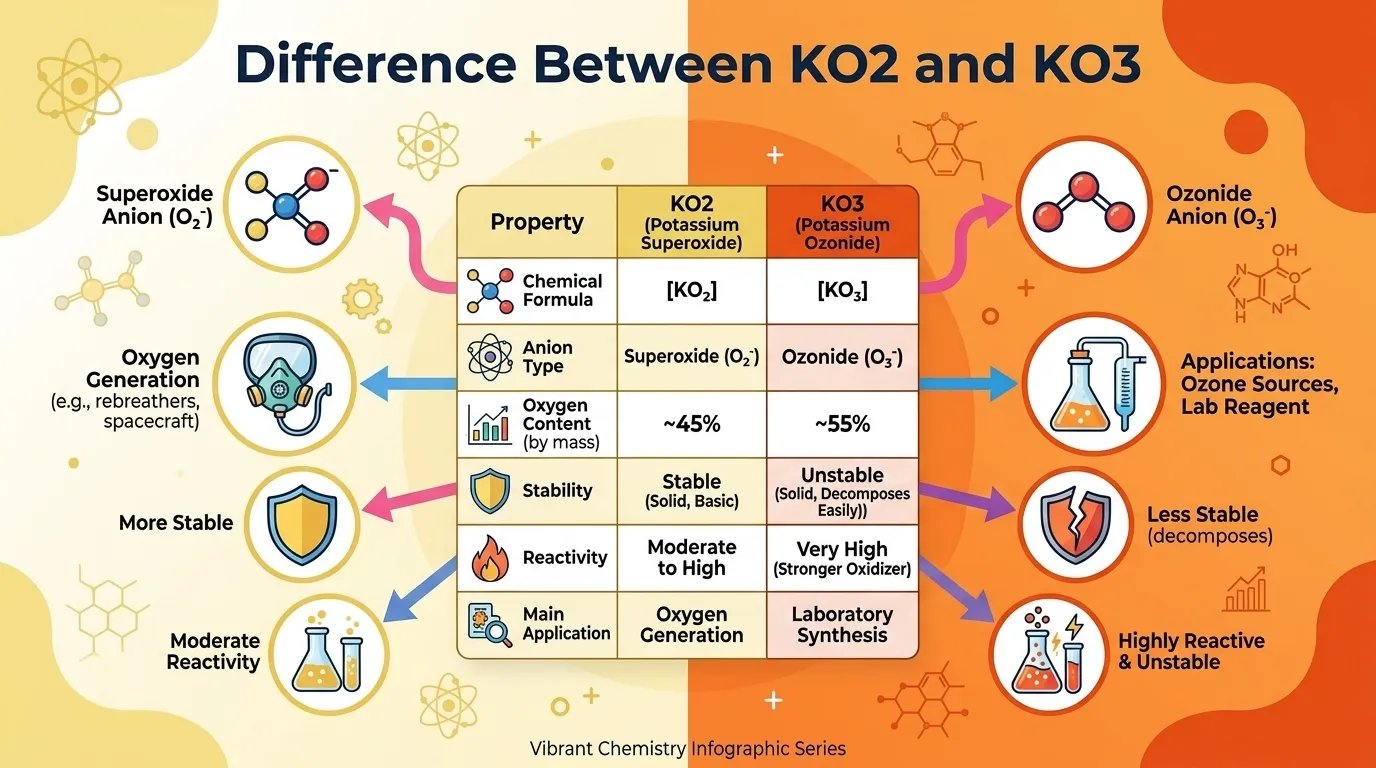
The main difference lies in their oxygen content and chemical nature. KO2 (potassium superoxide) contains one potassium atom and two oxygen atoms, while KO3 (potassium ozonide) contains one potassium atom and three oxygen atoms. This difference changes their stability, reactivity, and practical uses significantly.
Why Is Their Difference Important?
Understanding these compounds is important for both learners and experts. KO2 is widely used in oxygen generation systems, especially in closed environments like submarines or spacecraft. KO3, on the other hand, is less stable and mainly studied for research purposes.
For students, knowing the difference between them helps build a strong foundation in inorganic chemistry. For experts, it is crucial in designing systems for oxygen supply and studying reactive oxygen species. In society, these compounds contribute to safety systems, environmental protection, and scientific advancement.
Pronunciation of the Both
- KO2 (Potassium Superoxide)
- US: /poʊˈtæs.i.əm suːˈpɝː.ɑːk.saɪd/
- UK: /pəˈtæs.i.əm suːˈpɜː.ɒk.saɪd/
- KO3 (Potassium Ozonide)
- US: /poʊˈtæs.i.əm ˈoʊ.zə.naɪd/
- UK: /pəˈtæs.i.əm ˈəʊ.zə.naɪd/
Now that we know how to pronounce them, let’s move deeper into understanding their differences.
Difference Between KO2 and KO3
1. Chemical Formula
KO2 has two oxygen atoms, while KO3 has three.
- Example 1: KO2 = K + O2
- Example 2: KO3 = K + O3
2. Type of Compound
KO2 is a superoxide, KO3 is an ozonide.
- Example 1: KO2 used in oxygen masks
- Example 2: KO3 studied in labs
3. Stability
KO2 is more stable than KO3.
- Example 1: KO2 stored safely
- Example 2: KO3 decomposes quickly
4. Reactivity
KO3 is more reactive than KO2.
- Example 1: KO2 reacts slowly with water
- Example 2: KO3 reacts violently
5. Oxygen Release
KO2 releases oxygen steadily, KO3 releases rapidly.
- Example 1: KO2 in breathing devices
- Example 2: KO3 in experimental setups
6. Color
KO2 is yellow, KO3 is reddish.
- Example 1: KO2 appears pale yellow
- Example 2: KO3 shows orange-red tint
7. Usage
KO2 has practical uses, KO3 mostly research-based.
- Example 1: KO2 in submarines
- Example 2: KO3 in chemical studies
8. Structure
KO2 contains O2⁻ ion, KO3 contains O3⁻ ion.
- Example 1: KO2 simple structure
- Example 2: KO3 complex structure
9. Safety
KO2 is safer to handle than KO3.
- Example 1: KO2 used industrially
- Example 2: KO3 handled in controlled labs
10. Availability
KO2 is widely available, KO3 is rare.
- Example 1: KO2 used commercially
- Example 2: KO3 limited production
Nature and Behaviour of the Both
KO2 is relatively stable and reacts moderately with moisture, producing oxygen and potassium hydroxide. It behaves as a strong oxidizing agent but is manageable in controlled conditions.
KO3 is highly unstable and reactive due to the presence of the ozonide ion. It decomposes easily and can release oxygen rapidly, making it more dangerous and less practical for everyday use.
Why People Are Confused About Their Use?
| Feature | KO2 | KO3 | Similarity |
|---|---|---|---|
| Composition | K + O2 | K + O3 | Both contain potassium |
| Type | Superoxide | Ozonide | Oxygen-rich compounds |
| Stability | Stable | Unstable | Reactive nature |
| Use | Practical | Research | Scientific importance |
| Reactivity | Moderate | High | Oxidizing agents |
People often confuse them because both are potassium-oxygen compounds and appear similar in formulas.
Which Is Better in What Situation?
KO2 is better for practical applications. It is widely used in oxygen masks, submarines, and emergency breathing systems because it releases oxygen in a controlled way. It is stable enough for industrial use and safer to handle compared to KO3.
KO3 is better suited for research purposes. Scientists use it to study highly reactive oxygen species and chemical reactions. However, due to its instability, it is not suitable for everyday or industrial applications.
Use in Metaphors and Similes
Though scientific, they can be used metaphorically:
- “His energy was like KO3 powerful but unstable.”
- “Her consistency was like KO2 steady and reliable.”
Connotative Meaning
- KO2: Positive (stability, usefulness)
- Example: “A KO2-like system ensures reliability.”
- KO3: Negative/Neutral (instability, unpredictability)
- Example: “His mood swings were KO3-like.”
Idioms or Proverbs Related
No direct idioms exist, but adapted usage:
- “Handle it like KO3” (meaning: carefully)
- “Be as steady as KO2” (meaning: reliable)
Examples:
- “In crisis, be as steady as KO2.”
- “This situation must be handled like KO3.”
Works in Literature
- “Principles of Inorganic Chemistry” – Textbook, Various Authors, 20th century
- “Advanced Oxidation Processes” – Academic, Research Writers, 2000s
Movies Related (Scientific Context)
- “The Martian” (2015, USA) – Oxygen generation themes
- “Apollo 13” (1995, USA) – Survival and oxygen systems
FAQs
1. What is the main difference between both?
KO2 is a superoxide, while KO3 is an ozonide with higher reactivity.
2. Which is more stable?
KO2 is more stable than KO3.
3. Where is KO2 used?
In oxygen masks and submarines.
4. Why is KO3 unstable?
Due to the highly reactive ozonide ion.
5. Are both dangerous?
Yes, but KO3 is more dangerous due to higher reactivity.
How Both Are Useful for Surroundings
KO2 helps in maintaining breathable air in confined environments, making it highly useful for human survival systems. KO3 contributes to scientific understanding of reactive oxygen species, helping in research and innovation.
Final Words for the Both
KO2 represents stability and practicality, while KO3 represents high reactivity and scientific curiosity. Both are important in their own domains.
Conclusion
In conclusion, the difference between KO2 and KO3 lies in their composition, stability, and application. KO2 is widely used and practical, while KO3 is mainly limited to research due to its instability. Understanding the difference between both helps students and professionals grasp important chemical concepts and apply them in real-world situations. Ultimately, both compounds highlight the fascinating diversity of chemistry and its impact on modern life.

I am content creator and comparison blogger focused on analyzing key differences between terms, concepts, and ideas to deliver accurate, easy-to-understand information. So I decided to create a platform where these differences are explained in the simplest way possible.